Now imagine this – it was just an ordinary day in West Africa. Under the scorching summer sun, you were building yourself a hut with mud bricks made out of the dried ground. Life was peaceful until one night, when there was an unusually heavy rain. You, soaked in rainwater, sadly watched the mud being washed off your home, but were soon horrified – snake-like creatures were coming out of the bricks and dragging themselves down the walls! Now, don’t panic, because you just witnessed one of the most amazing animals on Earth – the lungfish, who have been asleep in your mud walls for months.
The African lungfish, genus Protopterus, are “living fossils” that have survived on the Earth for nearly 400 million years, through drastic environmental changes such as the drying of the Sahara. One of the main adaptations that allows lungfish to be the ultimate survivor is aestivation – dormancy in torrid seasons (as opposed to hibernation in winter), for up to an impressive 4 years.
Your encounter in Africa is not entirely hypothetical. And, believe it or not – people in Africa have even been digging lungfish out of dry mud for food.
So, how can an aquatic fish live in dry mud without any food or water, even sleeping in your wall for months without disturbance? To answer this question, curious scientists have been observing African lungfish since the 20th century.
In 1986, scientists from the University of Pennsylvania School of Medicine recorded the behavior of aestivating lungfish. Aestivation happens in dry seasons, as the retreating water leaves lungfish on moist mud. Without warning, lungfish starts to burrow a hole into the mud, and periodically resurfaces to take big gulps of air. It repeats this process until it’s lodged inside the hole in an upright U shape, with its mouth placed next to a thin channel in the ground for breathing.
By this time, lungfish has excreted enough tenacious mucus from its skin, which hardens to form a bed – a cocoon that envelops the lungfish’s entire body. Perhaps more unfortunate than starving, lungfish doesn’t get to turn in its sleep. It would remain this posture until enough water finally triggers its awakening.
With dissection and microscopy, scientists from the University of Genoa looked inside the amazing fish. To adapt to a new life underground, lungfish changes both the structure and function of its organs.
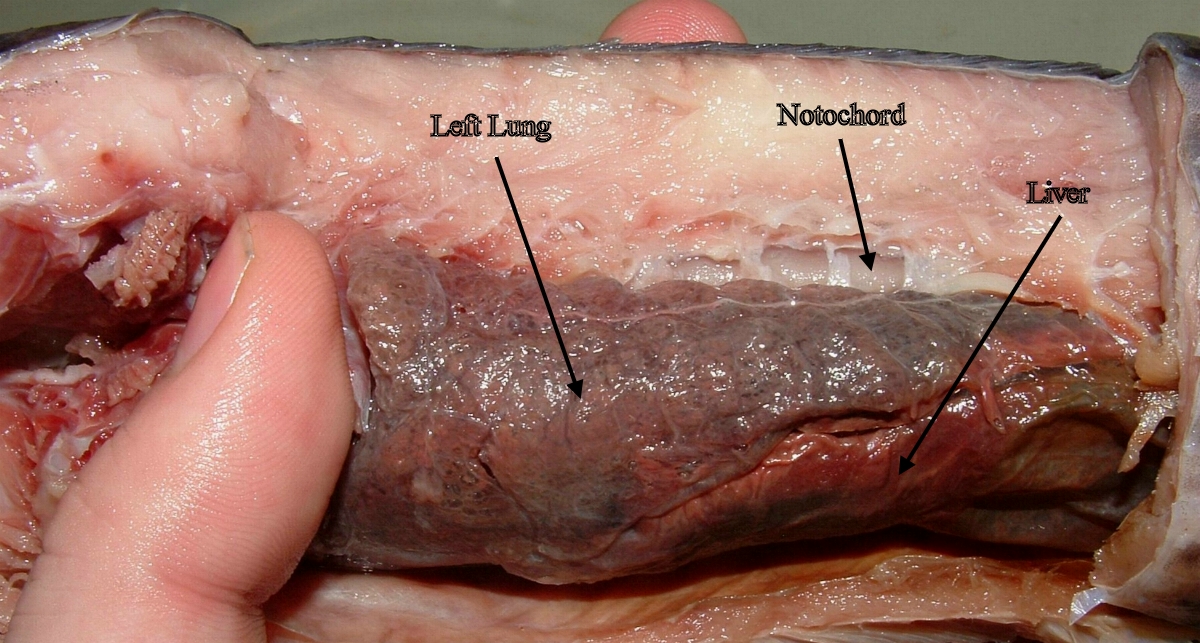
Most obviously, it transitions from water-breathing to air-breathing on land. True to its name, lungfish has both good old gills and lungs that resemble the lung types of primitive amphibians.
In water, lungfish’s gills have separated filaments with large surface area, increasing the gas exchange between blood and water. When on land, however, the aestivating fish excretes mucus that makes gill filaments stuck together, therefore non-functional.
On the contrary, the fish’s lungs that are thin bloodless threads in water are now swollen with alveoli – tiny “balloons” that transport gas into and out of the bloodstream. In this way, lungfish can breathe like a turtle.
In recent years, development of genetic tools has revealed more subtle changes inside the lungfish’s body. Through compiling genes and examining the expression levels of different DNAs, scientists from the National University of Singapore have found that lungfish adjusts its metabolism to resist stress and save energy.
For instance, apart from breathing, the aestivating fish shuts down another function of gills – excreting toxic waste as ammonia into the water. When on land, the lack of water makes this way of excretion impossible. To save energy, lungfish decreases the production of gill-proteins responsible for ammonia transport. It instead uses liver to convert ammonia into the less toxic urea.
The miracle of lungfish biology, however, does not stop with these clever mechanisms for dormancy. I can easily name a few more reasons for why lungfish is the coolest animal the Earth has ever seen.
(1) Lungfish has lots, lots of genes. The marbled lungfish from Africa (Protopterus aethiopicus) has the second largest genome – the total genetic material of an organism – found by scientists so far, amongst all species in the world. The only larger one belongs to a Japanese flower named Paris japonica.
(2) We are related. The similarity in physiology and gene sequences assigns lungfish as the closest living relative among fish of land vertebrates.
(3) Aestivation matters. From aestivation, hibernation, to tardigrades’ ability to shrivel up into a ball and survive in space, these common yet diverse forms of dormancy are called suspended animation. Scientists are now hypothesizing that there is a common genetic base for this ancient adaptation, which is likely to be also present in humans.
One essential question left unanswered, however, is the exact factors that trigger organisms into and out of dormancy. By potentially harnessing this “switch” of suspended animation, we can make ourselves survive in far harsher environments.
For patients awaiting organ transplants, this technology may be a life-savior; for astronomers, this raises hope to look for dormant life forms in outer space, or even put astronauts into dormancy for space exploration. Aestivation, as one unique form of dormancy found in an organism of evolutionary significance, is definitely worth more studying.

Astronauts entering “hypersleep” on the way to Saturn, from Nolan’s 2014 sci-fi epic Interstellar. Could this be true one day?
Before we can finally put cancer patients to a temporary sleep or send snoozing astronauts into deep space, let’s look at unexpected places for inspiration. Hidden underground, much more miracles of nature are there for us to see.
By: Z Zhang
References
Alekseev V.R., Sychev V.N., Novikova N.D. (2007). Studying the Phenomenon of Dormancy: Why it is Important for Space Exploration. Diapause in Aquatic Invertebrates Theory and Human Use. Monographiae Biologicae, (84), 207-214. Springer, Dordrecht. Retrieved from https://link.springer.com/chapter/10.1007%2F978-1-4020-5680-2_14
Chng, Y. R., Ong, J. L. Y., Ching, B., Chen, X. L., Hiong, K. C., Wong, W. P., . . . Ip, Y. K. (2017). Molecular characterization of three rhesus glycoproteins from the gills of the african lungfish, protopterus annectens, and effects of aestivation on their mRNA expression levels and protein abundance. PLoS One, 12(10), e0185814. Retrieved from http://dx.doi.org/10.1371/journal.pone.0185814
Fishman, A. P., Pack, A. I., Delaney, R. G., & Galante, R. J. (1986). Estivation in Protopterus. J. Morphol., 190(S1), 237–248. Retrieved from https://doi.org/10.1002/jmor.1051900416
Hiong, K. C., Ip, Y. K., Wong, W. P., & Chew, S. F. (2015). Differential gene expression in the liver of the african lungfish, protopterus annectens, after 6 months of aestivation in air or 1 day of arousal from 6 months of aestivation. PLoS One, 10(3), e0121224. Retrieved from http://dx.doi.org/10.1371/journal.pone.0121224
Sturla, M., Paola, P., Carlo, G., Angela, M. M., & Maria, U. B. (2002). Effects of induced aestivation in Protopterus annectens: A histomorphological study. J. Exp. Zool., 292(1), 26–31. Retrieved from https://doi.org/10.1002/jez.1139
Image Credits
Image 1: Mathae, Protopterus annectens at Paris Aquarium, https://commons.wikimedia.org/wiki/File:G%C5%91tehal-2.jpg
Image 2: Heuclin, D., West African lungfish (Protopterus annectens) buried in mud of dried river bed, Togo., //www.naturepl.com/stock-photo-nature-image01597946.html
Image 3: Unknown author, //lh3.googleusercontent.com/-FRv0ocgqMpU/WJw2hiUhcXI/AAAAAAABRtY/xxIvkiQzf5k/lungfish-52.jpg?imgmax=1600
Image 4: Mokele, Lungs of Protopterus dolloi, https://commons.wikimedia.org/wiki/File:Lungs_of_Protopterus_dolloi.JPG
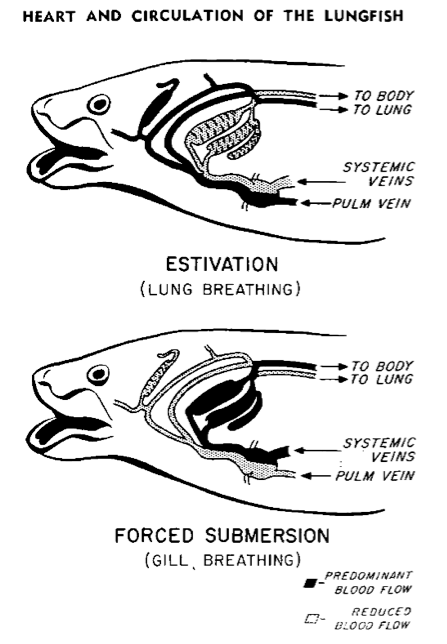
Image 5: Szidon, Jan P., Figure 16 from Heart and Circulation of the African Lungfish, https://www.ahajournals.org/doi/abs/10.1161/01.res.25.1.23
Image 6: Interstellar (2014), Romilly entering hypersleep on the way to Saturn, https://www.imdb.com/title/tt0816692/