Kailyn Valido
The majority of people dread the inevitability of aging, a genetically programmed process. It is evident that many wish that they could somehow rewind time to relive their 20’s and 30’s—a range of years where most people do not have wrinkles, graying hair, and frequent thoughts about the enigmatic nature of “The Great Unknown.”
What if one can extend this period of youthfulness and vivacity? What if one can delay this seemingly undesirable process of aging?
This is where the notion of telomeres comes into play.
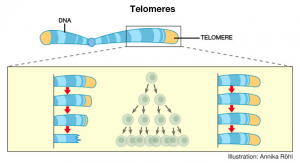
Telomeres are essentially repeated sequences of DNA at the end of eukaryotic chromosomes. These sequences are in the form of repeated TTAGGG bases on one strand of DNA and AATCCC bases on the complementary DNA strand. Analogous to the plastic tips on shoelaces, telomeres ultimately provide the chromosome with protection—up to a certain point.
Synthesized by the enzyme telomerase, telomeres get shorter every time a cell undergoes division. Because there is not enough telomerase for somatic—or body—cells, the gene for telomerase is inactive and telomeres consequently shorten. The shorter the telomere length, the lesser amount of times a cell can divide. Once the telomeres become too short, the cell can no longer divide, and so it ages and inevitably dies.
For this reason, telomere shortening has been linked to the aging process. Through his research, geneticist Richard Cawthon at the University of Utah discovered that shorter telomeres correlate with shorter lives. Working with a group of people older than 60 years old, he and his colleagues found that those who had significantly shorter telomeres were three times more likely to die from heart disease and eight times more likely to die from infectious disease.
The main issue with this finding, however, relates to the correlation versus causation argument, in which it is commonly believed that correlation does not equal causation. Scientists are currently unsure of whether telomere shortening is merely a sign of aging, such as wrinkles, or an actual contributing factor.
If telomere shortening is proven to be a definite cause of aging, this information would be an immense breakthrough in the medical field. Scientists may be able to extend human lifespan greatly by somehow devising a mechanism to restore or preserve telomere length, allowing cells to divide more and not become senescent.
In a small-scale scientific study at the University of California at San Francisco, scientists wanted to research if telomere length can be affected by certain lifestyle changes. In this study, which began in 2008, 35 men who had early-stage prostate cancer were closely monitored. Ten men altered their lifestyle habits by having a plant-based diet, moderate exercise six days a week, and stress reduction exercises. Additionally, they attended a weekly support group. The 25 other men who had early prostate cancer were not asked to make any major lifestyle alterations.
After five years of surveillance, the 10 men with the lifestyle changes had an increase in telomere length by about 10 percent. In contrast, those 25 men who did not change any lifestyle habits had significantly shorter telomeres, about 3 percent shorter when the study was concluded.
This pilot study has major implications. Because of the small size of the study, however, the legitimacy of this research finding needs to be confirmed by more large-scale studies to assess the sample size and find repeatability in results. The scientists at UCSF hope to inspire larger research studies for confirmation.
The lead scientist in this study, Dean Ornish, MD, powerfully states, “Our genes, and our telomeres, are not necessarily our fate…[The] findings indicate that telomeres may lengthen to the degree that people change how they live.”
If scientists can preserve telomere length, one might be wondering then, will humans be able to attain that much desired Fountain of Youth and ultimately become immortal like many science fiction movies portray? Very unlikely.
Although the concept of telomere lengthening and preservation can potentially postpone aging, unnaturally active telomerase and incessant division of cells may be problematic. Cancer cells, for instance, escape their death by having a consistently active telomerase, preventing telomeres from shortening. This, in turn, allows these cells to grow an abnormal amount of times and form tumors. Scientists are aware that replicative aging of cells occurs to combat against these malignancies.
This complex part of telomerase and its product, telomeres, is what is still confounding experts today. If telomerase can be systematically lengthened to extend one’s lifespan, would that increase that person’s risk of cancer? Questions similar to these and their respective multifaceted issues are currently being explored. Perhaps one cannot delay the aging process without certain repercussions, such as a higher chance of having cancer.
Scientists, however, are still hopeful. Many are attempting to utilize the notion of telomeres as a target for cancer treatment. If they can find a way to shorten cancer cell telomeres and allow these cells to age and die, it can be revolutionary.
Some researchers have already started testing this in the lab with prostate cancer cells, but results had unfortunate consequences, such as significant impairment of both fertility and production of immune cells. Knowledge is still being gathered about how differentiation in normal cells and cancer cells can be related to telomeres and the enzyme telomerase.
Despite scientists’ extraordinary efforts in fighting against the aging process by manipulating and observing telomeres, the only seemingly possible result is to delay aging rather than terminating the process altogether. In addition to the preservation of life, research on telomeres could be a key to unlocking the cure to cancer— a colossal discovery that would prevent a lot of emotional, physical, and mental suffering in the world.